Medovate has announced a newly formed partnership with Konica Minolta Healthcare Americas, Inc. to promote best practice within regional anaesthesia across the United States and the Middle East.
The partnership will highlight the provision of Ultrasound Guided Regional Anaesthesia’ and how the revolutionary SAFIRA® (SAFer Injection for Regional Anaesthesia) can be used in conjunction with Konica Minolta Healthcare Americas’ Inc. range of pioneering ultrasound solutions.
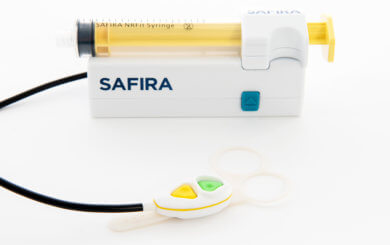
SAFIRA® is a Class II medical device set to transform regional anaesthesia around the world by making it a one-person procedure. It puts control of the injection in the hands of the anaesthetist freeing up their assistants to carry out other tasks. The device also helps to improve patient safety by reducing the risk of nerve damage as it helps to prevent injection above 20psi.
The introduction of ultrasound guidance in regional anaesthesia was first explored by anaesthetists in the mid-1990s and since then technology advancements have led to an increase in ultrasound byanaesthetists.
Ultrasound guidance offers a range of important practical advantages for nerve blockade such as visualisation of the anatomy and more informed guidance for the needle pathway avoiding structures that might be damaged by the needle. It also reduces the likelihood of inadvertent needle trauma to unintended structures and ensures the continued visualisation of the local anaesthetic solution.
Together the two technologies present a best practice solution in delivering regional anaesthesia.
Speaking about the partnership, Chris Rogers, Sales and Marketing Director at Medovate, said:
“We are excited to be partnering with Konica Minolta Healthcare Americas, a world leader in ultrasound technology. The company has a strong focus on creating value through innovation – an ethos that fits with Medovate’s core focus on realising the benefits of clinician-led innovation. We are looking forward to raising awareness of ultrasound guided techniques for regional anaesthesia.”
Recent publications, including a joint statement by the American Society of Regional Anesthesia and Pain Management (ASRA) and the European Society of Regional Anaesthesia and Pain Therapy (ESRA), have recommended regional anaesthesia be considered whenever surgery is planned for a suspected or confirmed COVID-19 patient. This is because it preserves respiratory function and avoids aerosolisation and the potential for transmission of COVID-19 compared to general anaesthesia.
Eric Sumner, Executive Vice President Ultrasound Sales at Konica Minolta, said:
“Medovate brings a unique combination of clinical innovation and expertise through its association with the United Kingdom’s National Health Service and we are proud to partner with them to advance regional anaesthesia. Our companies have a shared vision to promote safer solutions that deliver clinical efficiency, simplify use and advance better outcomes for patients.”
Joan Toth, Senior Product Marketing Manager for Konica Minolta Healthcare Americas Inc., added:
“Our companies are committed to furthering the use of regional anesthesia in the US. Through Konica Minolta’s UGPro solution, we’ll provide the latest ultrasound technology and procedural innovations, such as SAFIRA®, along with hands-on education to help anaesthetists offer this important alternative to their patients. Medovate’s clinical knowledge and experience are invaluable and we are excited to partner with them.”
News & Analysis